Dipole moment definition chemestry12/31/2023  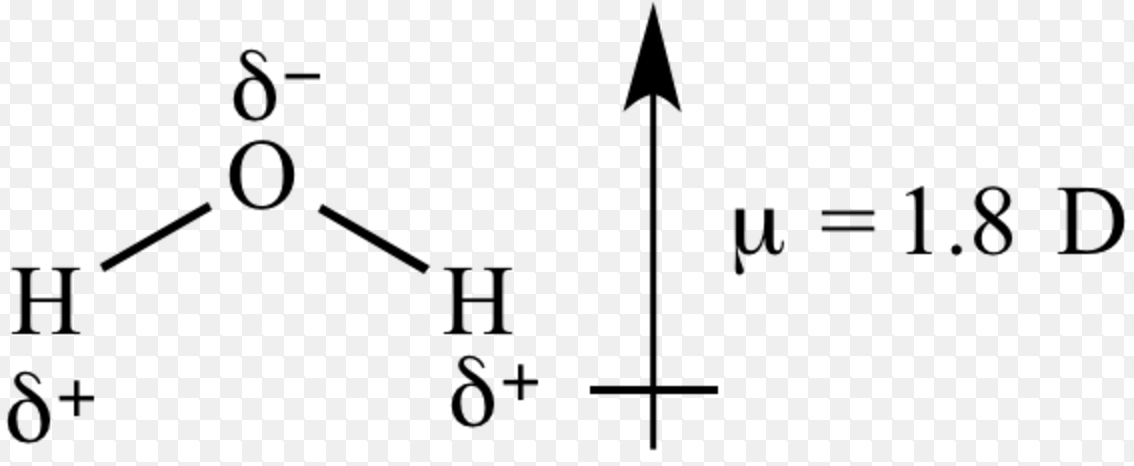
It is also found in particle physics in the measurement of possible dipole moments of electrons and neutrons for example. Usually used for atoms and molecules, the notion of dipole moment is used to describe the electric fields of such systems as well as Van der Waals forces. 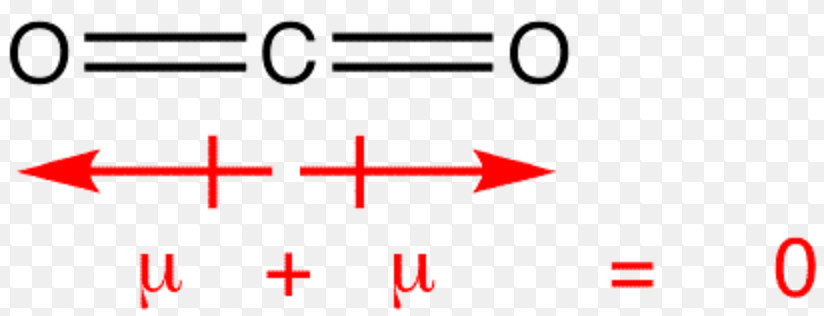
m) but it is often also expressed in debyes (D).The dipole moment is expressed in coulomb A quantitative measurement of the bonds polarity or molecules polarity is termed as dipole moment. Molecular geometry, also known as the molecular structure, is the three-dimensional structure or arrangement of atoms in a molecule. More generally, a dipole moment is the sum of n vectors P i=q ir i where q i and r i are respectively the values of n charges q i having n position vectors r i. The vector d is oriented from the negative charge to the positive charge. Dipoledipole forces occur between molecules with permanent dipoles (i.e., polar molecules). The dipole moment is a measure of how polar a bond is The direction of the dipole moment is shown by the following sign in which the arrow points to the. A dipole moment is a vector generally noted P or D.įor two identical charges of opposite sign +q and -q, separated by a distance d, the dipole moment is described by the vector P=qd. The simplest dipole is therefore a pair of two charges of opposite sign separated by a finite distance. This is an unequal distribution of electrical charges where the barycentre of the positive charges does not coincide with that of the negative charges. The notion of dipole moment in physics and chemistry is based on the existence of electrostatic dipoles. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
